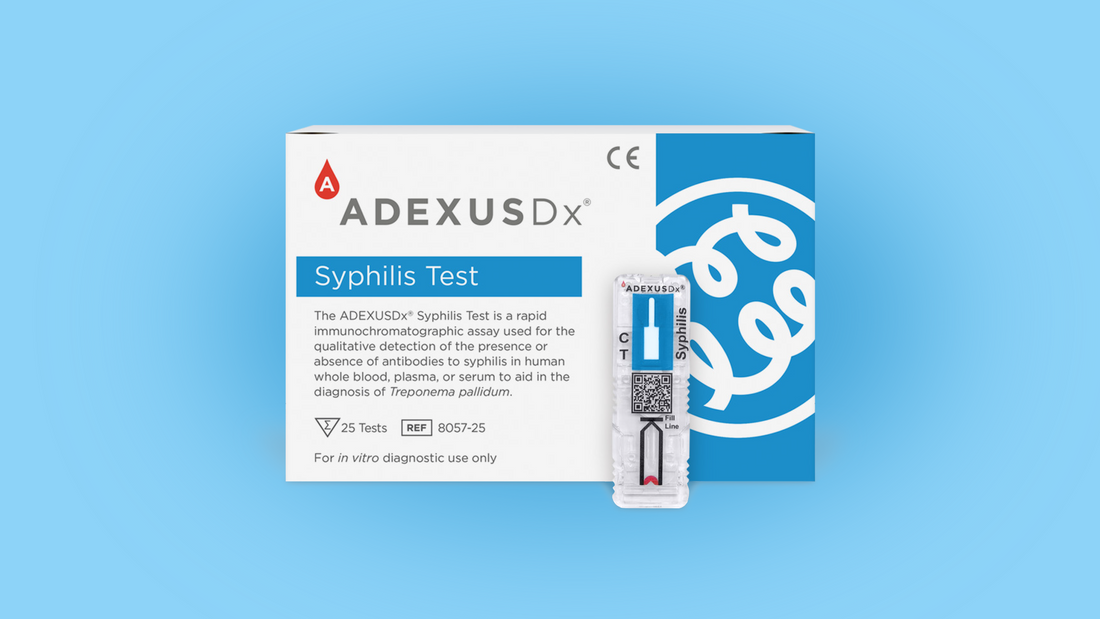
SPRINGDALE, ARK. – March 25, 2022 – NOWDiagnostics was granted CE Mark for the ADEXUSDx® Syphilis rapid test. This treponemal assay requires only 1 drop of whole blood to perform and yields a laboratory quality result in just 15 minutes. The test has 100% sensitivity for all stages of syphilis and 99% specificity. In a seroconversion study, the test detected syphilis as early as or earlier than 12 other syphilis assays. Syphilis is a sexually transmitted disease caused by the bacterium Treponema pallidum. It is transmitted by direct contact with a syphilitic sore, known as a chancre. Syphilis typically follows a progression of stages (primary, secondary, latent, tertiary) with varying symptoms and duration. Without treatment, the infection can be fatal or spread to the brain and nervous system resulting in neurosyphilis and be marked by a variety of symptoms including, but not limited to, headache, paralysis, and dementia. Syphilis can also spread to the eye resulting in ocular syphilis, marked by changes in vision and even blindness. Women with syphilis can transmit the infection to their babies in utero, resulting in low birth weight babies, and/or early or stillborn deliveries. Untreated babies can become developmentally delayed, have seizures or die.
The rate of syphilis continues to rise globally, with 7M new syphilis infections reported in 2020. The ADEXUSDx® Syphilis rapid test is an important new diagnostic tool to aid in early detection and prevent the spread of this deadly disease. The ADEXUSDx® Syphilis rapid test is now available for use in Europe and other countries reliant upon CE Mark. Customers may contact cc@nowdx.com to place orders today.